
When smoked, cocaine reaches the brain in 15 s. Write the melting point range of caffeine. It vaporizes at the temperature of a burning cigarette. Caffeine C8H10N4O - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more. Draw out the full structure of caffeine, showing all the bonds and lone pairs. The non-bonding electrons on each atom are present as lone pairs. Identify the location of all lone pairs in this compound: H3C CH3 N N N N. The space-filling model for caffeine is shown in Figure 3. Crack cocaine is more volatile than cocaine hydrochloride. 1.56 Below is the structure of caffeine, but its lone pairs are not shown. The name acetaminophen, which resembles 'aceto', 'amine', and 'phenol', which suggests that it looks like an acetoamide and phenol, which it does: 1) London Dispersion London dispersion is the weakest intermolecular force, and it is regarding an induced dipole. of caffeine: H O N C Complete a Lewis structure for caffeine in which all. Caffeine has 3 delocalised lone pairs of electrons on 3 nitrogens and only one N has electrons pair away from the aromatic r View the full answer. The Lewis structure helps us identify the bond pairs and the lone pairs. 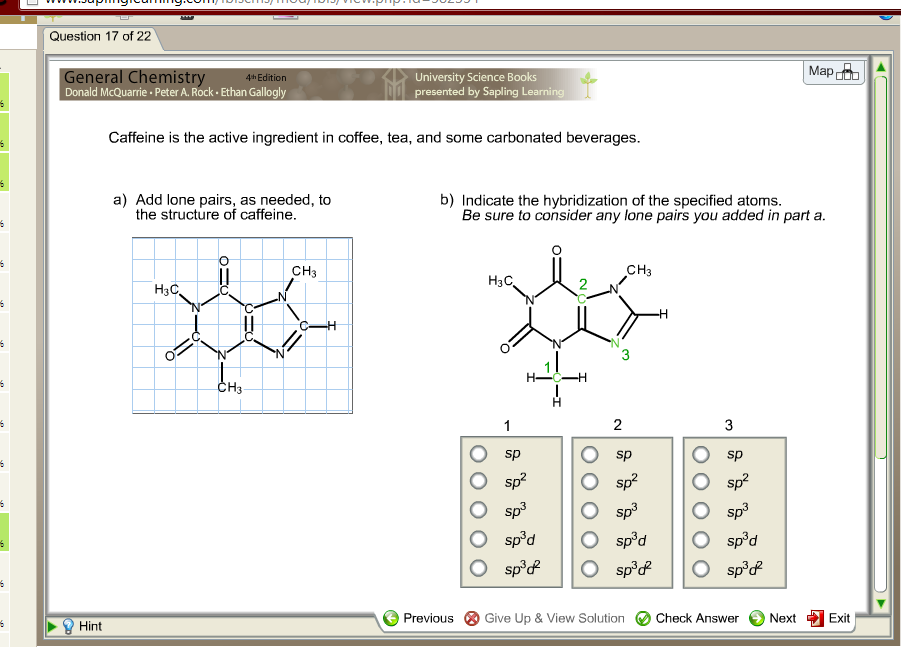
Although the Lewis theory does not determine the shapes of molecules, it is the first step in predicting shapes of molecules. The caffeine molecule contains a total of 25 bond(s) There are 15 non-H bond(s), 7 multiple bond(s), 2 double bond(s), 5 aromatic bond(s), 1 five-membered ring(s), 1 six-membered ring(s), 1 nine-membered ring(s), 1 urea (-thio) derivative(s), 1 imide(s) (-thio) and 1 Imidazole(s).\nonumber \]īecause it is soluble in water, cocaine hydrochloride is readily absorbed through the watery mucous membranes of the nose when it is snorted. Science Chemistry The structure of caffeine, present in coffee and many soft drinks, is shown here. Therefore, add lone pairs and multiple bonds to the structure below to give. Introduction To determine the shapes of molecules, we must become acquainted with the Lewis electron dot structure. 
How many lone pairs are in a caffeine molecule? The important thing to remember is that double bonds are comprised of 1 sigma bond and 1 pi bond and triple bonds are comprised of 1 sigma bond and 2 pi bonds. 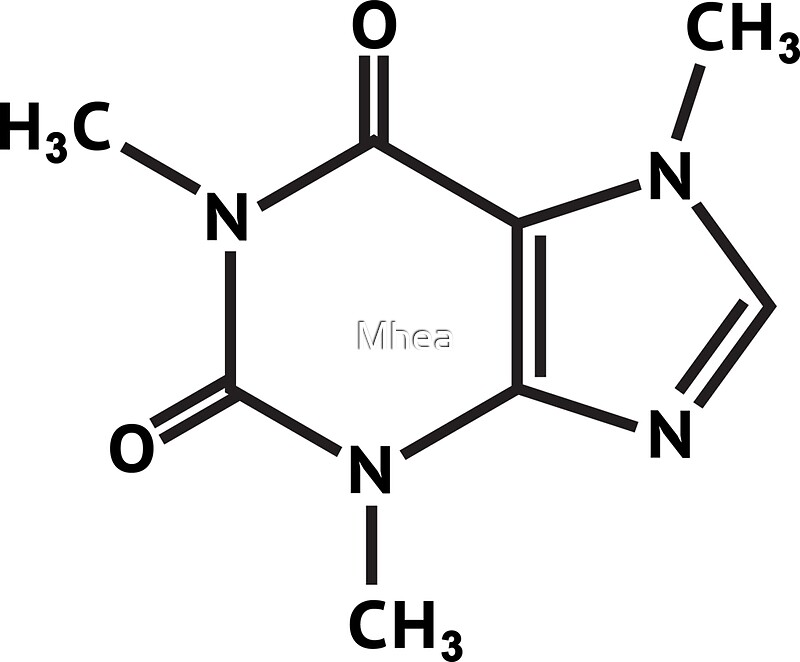
Every time you are asked to determine the number of pi or sigma bonds in a compound, you must draw its Lewis structure. Therefore, there are 8 lone pairs in caffeine. Oxygens with two bonds and a full octet will have two lone pairs, while nitrogens with three bonds and a full octet will have one lone pair. Suggested solution Thymine, a pyrimidine, has an alkene and lone pair. The elements of interest in caffeine for lone pairs are nitrogen and oxygen uncharged carbons will have no lone pairs. Me Me O N N Me N O HN N N H O Me thymine caffeine Purpose of the problem Revision. Read also : How does virtue ethics differ from deontology? How many lone pairs can you have?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
